MemoPart™ VSD is a percutaneous transcatheter device designed for the closure of ventricular septal defects (VSD). MemoPart™ VSDO includes four different types, divided into muscular VSDO and membranous VSDO.
- C/Duero, 37 bis, local 15, 28670 - Villaviciosa de Odón, Madrid
- info@cathmedical.com
Phone
+34 91 604 40 88
MEMOPART ™
Memopart™ are structural heart closure devices from Lepu Medical, marketed exclusively in Spain by our company.






Memopart ™
Valorado con 5 de 5
Top-Rated Products
Patent Foramen Ovale (PFO)
MemoPart™ PFO is a percutaneous transcatheter device intended for the closure of Patent Foramen Ovale (PFO) in patients with a history of stroke or transient ischaemic attacks (TIA).

Valorado con 5 de 5
PFO
Atrial Septal Defect (ASD)
MemoPart™ ASD is a percutaneous transcatheter device intended for the closure of atrial septal defects (ASD). It is also available for patients requiring fenestrated closure.

Valorado con 5 de 5
ASD
Ventricular Septal Defect (VSD)

Valorado con 5 de 5
VSD
Patent Ductus Arteriosus (PDA)
MemoPart™ PDA is a percutaneous transcatheter device intended for the closure of patent ductus arteriosus (PDA). The retention skirt ensures secure positioning within the ductal bulb.

Valorado con 5 de 5
PDA
Plug
The MemoPart™ plug is a percutaneous transcatheter device intended for the closure of a wide range of veins and arteries, reducing or eliminating blood flow by occluding the target vessel. The vascular plug can be used in patients with clinical symptoms associated with abnormal blood vessels, such as:
Aortopulmonary collaterals
Arteriovenous malformation (AVM)
Surgical aortopulmonary shunts
Anomalous venovenous connections
Arteriovenous fistulas
Peripheral vessels

Valorado con 5 de 5
PLUG
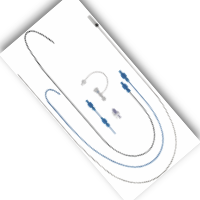
Delivery (Sistema de Entrega)
Two different sheath lengths are available for adults and paediatric patients.
The anti-kink delivery system is also available for VSD occluders.
The delivery system includes: Loader (1), Sheath (1), Dilator (1), Push Cable (1), Y-Connector with extension (1)
COMPACT DISCS
A: Long sheath
B: Dilator
C: Push cable
D: Y-connector with tube
Valorado con 5 de 5
ODS
China’s #1 Selling Product
1
Trusted by Professionals for More Than 12 Years
12
35% Share of the Chinese Market
35
140,000+ Cases Successfully Implanted Worldwide
140
MemoPart™

What is MemoPart™
MemoPart™ devices from Lepu Medical form a family of self‑expanding transcatheter occlusion systems designed for minimally invasive closure of a variety of congenital and structural heart defects. These include closure devices for Patent Foramen Ovale (PFO), intended to seal paths associated with stroke or transient ischaemic attacks; Atrial Septal Defect (ASD) occluders for the correction of atrial septal defects; Ventricular Septal Defect (VSD) occluders available in muscular and membranous types; and Patent Ductus Arteriosus (PDA) occluders for percutaneous closure of persistent ductus arteriosus. Each device typically features a self‑expanding double‑disc or mesh structure tailored to the specific anatomy of the defect, aiming to provide secure occlusion, high biocompatibility and visibility under fluoroscopy during implantation.
The comprehensive range of MemoPart™ solutions offers clinicians a versatile set of options for addressing a broad spectrum of structural cardiac abnormalities with minimally invasive techniques, supporting improved procedural outcomes and expanding the treatment possibilities for patients with congenital or acquired cardiac conditions
OCCLUDERS
Available with or without plug
Available to all types of MemoPart™
The physical technique applied to the device promotes earlier endothelialisation and improved biocompatibility.
The plug-free design is available for most occluders to promote earlier and improved endothelialisation.

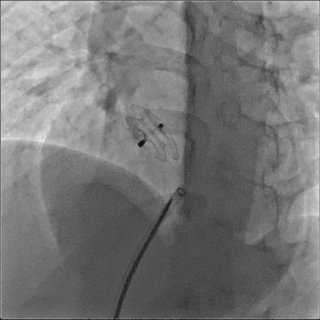

Occluders
Memopart ™
Occluders and vascular plugs are made from pre-oxidised nitinol wires, braided into a highly functional and elastic wire mesh, providing a high level of anatomical conformity. The short top plug design and screw housing are made of pre-oxidised stainless steel. The thickness of the individual wire varies according to size and type to ensure:
Functional elasticity
Enhanced shape memory
Easy positioning and deployment
High biocompatibility
Rapid endothelialisation
Minimized nickel release
The patch covering inside the discs and the connecting waist is made of PET, secured with a suture wire to ensure fast and effective endothelialisation.

Special Heat Treatment of Nitinol Wire
The oxidation process creates a compact and uniform TiO₂ surface on the nitinol wire.
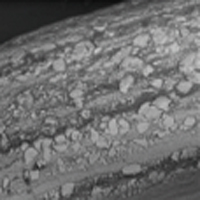
Excellent Haemocompatibility
Titanium oxide films provide the occluder with excellent haemocompatibility after implantation.

Long-Term Safety
The oxide layer effectively prevents the release of nickel ions (50% less than occluders without oxide coating), ensuring improved and earlier endothelialisation.

